
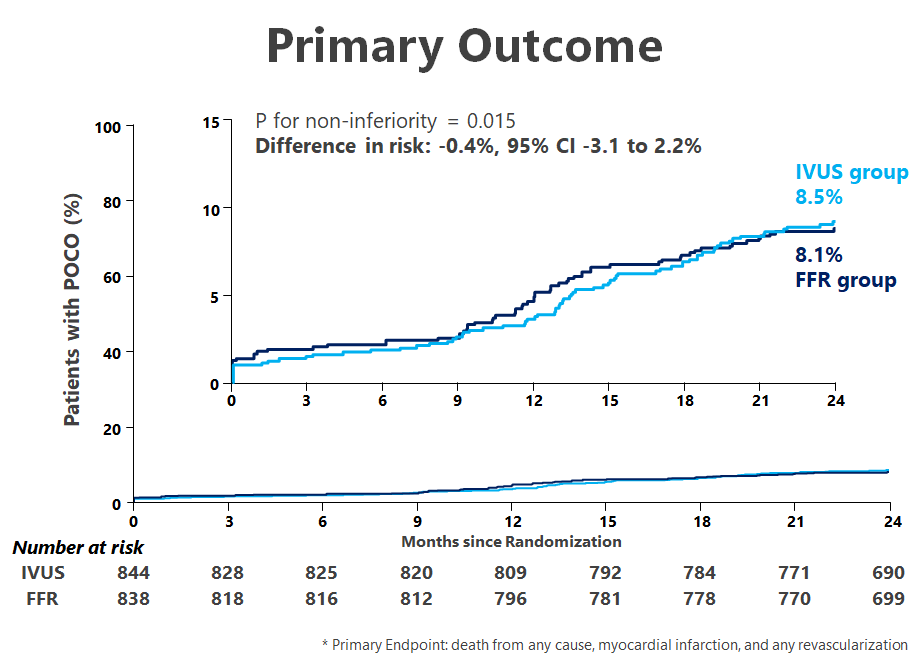
FLAVOUR Study, a multi-center international clinical trial, showed that in patients with intermediate stenosis, fractional flow reserve (FFR) guidance was noninferior to intravascular ultrasonography (IVUS) guidance in terms of the composite of death, myocardial infarction, or revascularization (8.1% vs. 8.5%, P=0.01 for noninferiority) at 24 months.
The study was led by Prof. WANG Jian'an, chair of SAHZU Heart Center and Head of SAHZU, and Prof. Bon-Kwon Koo, director of Cardiovascular Center and Chair in Cardiology Division of Seoul National University Hospital. The results were published on the New England Journal of Medicine (NEJM) on Sep. 1 Beijing Time.
For patients with intermediate coronary stenosis, cardiologists often use FFR to evaluate the potential of an ischemia-induced stenosis and whether percutaneous coronary intervention (PCI) is needed.
Meanwhile, they also use IVUS to select the appropriate stent size and to evaluate whether the vessel is optimally stented after procedure.
Therefore, combining FFR to make PCI decision and IVUS to improve stent implantation is considered as the best strategy.
However, many cardiac catheterization laboratories are equipped with only one of them. Given the lacking data regarding the difference between FFR- or IVUS-guided strategies, a head-to-head comparison between the two strategies was investigated in the study.
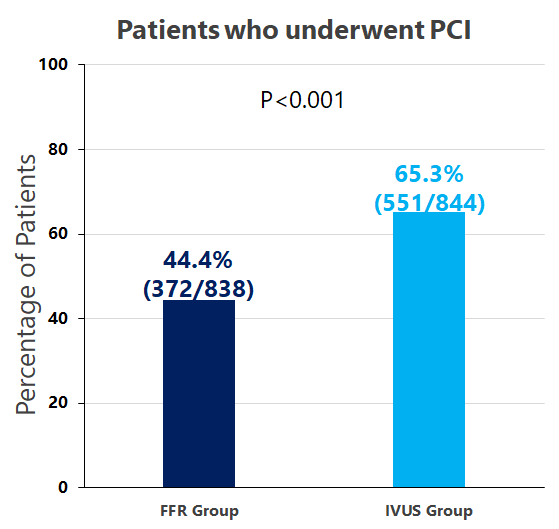
FLAVOUR Study is a prospective, randomized, open-label trial that is conducted on 1,682 patients with intermediate stenosis from 18 Chinese and Korean medical centers.
According to the prespecified criteria for revascularization, 44.4% of patients in the FFR group and 65.3% of those in the IVUS group underwent PCI. At 24 months, a primary-outcome event had occurred in 8.1% of the patients in the FFR group and in 8.5% of those in the IVUS group with non-inferiority p=0.01.
Accordingly, Prof. WANG Jian'an, Prof. Bon-Kwon Koo and their research team conclude that, in patients with intermediate stenosis, FFR guidance was noninferior to IVUS guidance with respect to the primary outcome, which was defined as a composite of death, myocardial infarction, or revascularization at 24 months.
Jane Leopold, Deputy Editor of NEJM and Associate Professor of Medicine at Harvard Medical School, wrote an editorial for the paper and spoke highly of the trial as "of immediate relevance to cardiologists and cardiac catheterization laboratories with only one of the two devices, either FFR or IVUS," and she looked forward to future studies that assess either the use of FFR or IVUS alone vs. the combination strategy, or the applicability of these results to patients with higher risks.
Prof. WANG Jian'an is the lead corresponding author of this publication. Prof. HU Xinyang is the co-first author.
Except for SAHZU, the other 8 participating Chinese centers are the First Affiliated Hospital of Wenzhou Medical University, the Second Affiliated Hospital of Wenzhou Medical University, Ningbo First Hospital, Hangzhou First People's Hospital, the Affiliated Hospital of Hangzhou Normal University, Zhejiang Hospital, Wenzhou People's Hospital and the Affiliated Hospital of Ningbo University School of Medicine.
Editted and Translated from: https://mp.weixin.qq.com/s/JLt1ajtH4Vw3ELFIjIBMCg
Author: LI JING | Reviewer: ZHANG JINLONG | Editor: LI JING | Source: NEJM医学前沿 | Date:2022-09-02 | Views:![]()
![]()
![]()
![]()