

Lung cancer is the leading cause of cancer-related deaths worldwide, with non-small cell lung cancer (NSCLC) accounting for 85% of all cases. Overall survival (OS) at 5 years after surgery remains poor due to high incidence of postoperative recurrence and metastasis.
To improve the efficacy of the treatment for NSCLC, the outcome of two vs three cycles of neoadjuvant sintilimab combined with chemotherapy in resectable stage IB-IIIA NSCLC patients was recently evaluated by Prof. QIU Fuming, a medical oncologist at SAHZU, and Prof. FAN Junqiang, a cardiac surgeon at SAHZU (NCT04459611).
According to their study, increasing the number of cycles of neoadjuvant treatment from two to three leads to a numerical improvement in major pathological response (MPR) with good tolerability.
Their research finding entitled Two vs three cycles of neoadjuvant sintilimab plus chemotherapy for resectable non-small-cell lung cancer: neoSCORE trial was published in Signal Transduction and Targeted Therapy (IF=38) on April 10. (View the article: https://www.nature.com/articles/s41392-023-01355-1 )
Between July 2020 and September 2021, 60 eligible patients of enrolled 64 patients were randomized by the study. 91.7% (55/60) of the 60 patients received surgical treatment. Two patients (6.9%) in three-cycle group had pneumonectomy.
MPR was observed in 19 patients among the 55 patients who underwent surgery, with an MPR rate of 26.9% in the two-cycle group and 41.4% in the three-cycle group. In a planned subgroup analysis of MPR according to baseline characteristics, no significant differences were observed between subgroups.
In the study, squamous NSCLC cases present a higher MPR rate than non-squamous NSCLC cases. The squamous subtype was proved to be significantly related to MPR in the multivariate logistic regression.

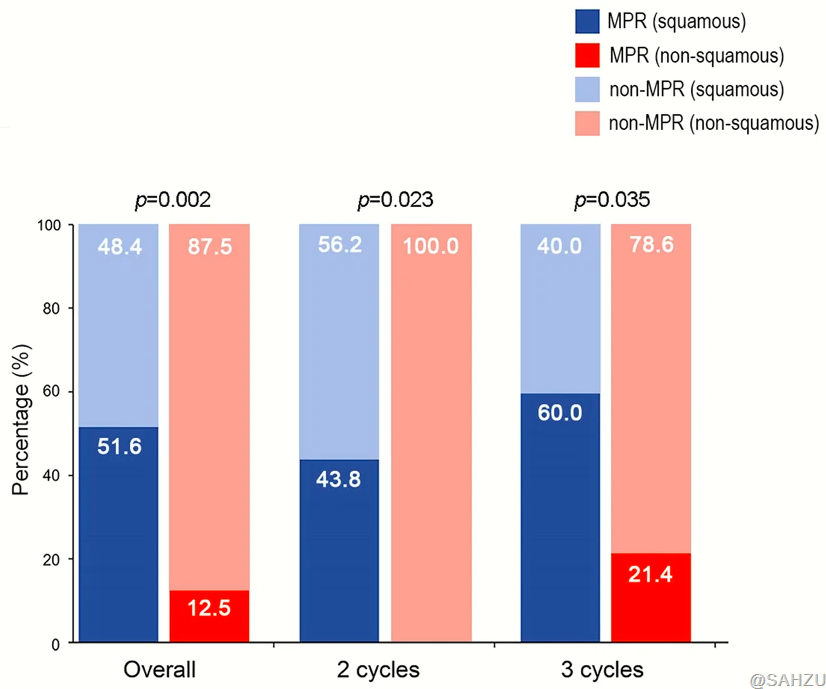
According to the article authors, this is the first randomized trial to assess the efficacy and safety of different cycles of neoadjuvant immuno-chemotherapy for resectable NSCLC.
SAHZU researchers believe the study provides valuable insight into the number of cycles of neoadjuvant treatment plus chemotherapy. By increasing the cycle number from two to three, MPR rate could be improved with good tolerability.
To continue assessing the relation between prolonged treatment cycle and improved MPR, SAHZU researchers are planning to conduct a phase 3 clinical trial to compare three vs. four cycles of neoadjuvant immune-chemotherapy in resectable squamous NSCLC patients.
Author: LI JING | Reviewer: SAHZU MEDICAL ONCOLOGY | Editor: LI JING | Source: iNATURE | Date:2023-04-25 | Views:![]()
![]()
![]()
![]()
88 Jiefang Road,Shangcheng District Hangzhou,China, 310009
1511 Jianghong Road,Binjiang District Hangzhou,China, 310014
300 Yuanju Road, Shangcheng DIstrict, Hangzhou
456 Qidi Road, Xiaoshan District, Hangzhou, China
1 Xihu Avenue, Shangcheng District, Hangzhou
Zijingang Campus of Zhejiang University, 866 Yuhangtang Road, Xihu District, Hangzhou
Please call +86-571-8971 3988 (8am-5pm, Monday through Friday)
Make an appointment online

The Second Affiliated Hospital
Zhejiang University School of Medicine
88 Jiefang Road, Hangzhou, China
+86-571-8731 5108
iao_sahzu@zju.edu.cn
The Needs of Patients and Customers Come First.
The Second Affiliated Hospital Zhejiang University School of Medicine
All Rights Reserved.

